Ascher Group

"Our group is interested in developing and experimentally validating novel computational methods to exploit this data, enhancing the impact of genome sequencing, structural genomics, and functional genomics on biology and medicine." - David Ascher
Research
On-going technological advancements have led to dramatic increases in the amounts of biological data being generated. Along with the evolution of high performance computing and computational tools, this has provided us with a wealth of information, analytical power and the opportunity to investigate fundamental health and biotechnological problems of a different magnitude and kind, complementary to and able to guide conventional approaches.
One of our main areas of interest is in the development of predictive and analytical tools and databases to investigate and understand the relationship between protein sequence, structure and function and phenotype, allowing us to gain unique insights into:
- The molecular basis of genetic diseases, including cancer;
- Understanding the molecular mechanisms behind drug resistance, to guide personalized patient treatment and the development of resistance resistant drugs;
- Evolutionary insights derived from the analysis of protein structure and function;
- Small molecule activity and toxicity as an aid to the design of novel drugs.
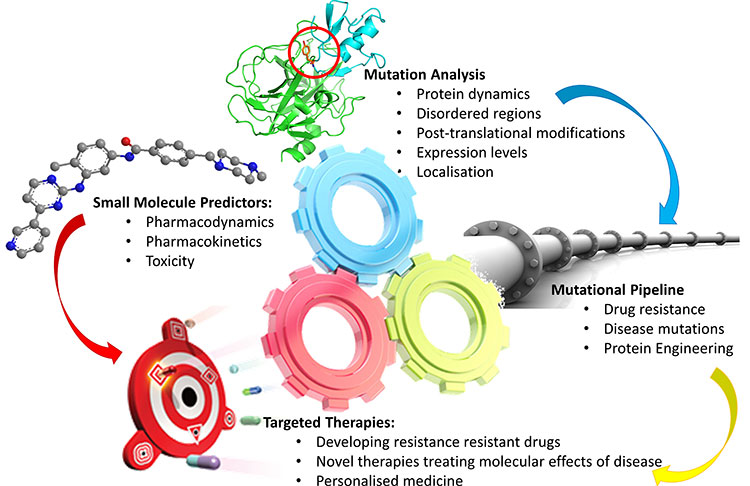
We have developed and host a wide range of widely used and freely-available tools, including:
- Arpeggio: Calculation and visualisation of all molecular interactions.
- MTR Viewer: Quantification of the extent of localized purifying selection in protein-coding sequences.
- mCSM-Stability: Predicting effects of mutations on protein stability.
- DUET: An integrated method for predicting effects of mutations on protein stability.
- SDM2: An optimised knowledge based method for predicting effects of mutations on protein stability.
- DynaMut: Analysis of protein dynamics and predicting the impact of mutations on protein conformation, flexibility and stability
- mCSM-PPI: Predicting effects of mutations on the affinity of protein-protein interactions.
- mCSM-AB: Predicting effects of mutations on antibody-antigen binding affinity.
- mCSM-NA: Predicting effects of mutations on the affinity of protein-nucleic acid interactions.
- mCSM-NAv2: Optimised predictions of the effects of mutations on the affinity of protein-nucleic acid interactions.
- mCSM-lig: Predicting effects of protein mutations on affinity for small molecules.
- CSM-lig: Predicting the protein binding affinity of small molecules.
- Kinact: Identification of protein kinase activating mutations.
- pkCSM: Predicting small molecule pharmacokinetic and toxicity properties.
- Platinum DB: Structural database of experimentally measured effects of missense mutations on protein-ligand complexes.
- TROMBONE DB: Optimisation of Botulinum and Tetnus neurotoxins for medicinal purposes.
- Symphony DB: Classification of VHL missense mutations according to risk of clear cell Renal carcinoma.
 Group Members
Group Members
Dr David Ascher, Group Leader
Postdoctoral Scientist
Dr Douglas Pires (Fiocruz-Minas)
Masters Students
Eleni-Maria Michanetzi, Masters Student (Melbourne)
Vasishth Sidarala, Masters Student (Melbourne)
Willy Cornelissen, Masters Student (UFMG, Brazil)
Amanda Tábita da Silva Albanaz, Masters Student (UFMG, Brazil)
PhD Students
Carlos Rodrigues, PhD Student (Melbourne)
Malancha Karmakar, PhD Student (Melbourne)
Michael Silk, PhD Student (Melbourne)
Stephanie Portelli, PhD Student (Melbourne)
YooChan Myung, PhD Student (Melbourne)
Liviu Copoiu, PhD Student (Cambridge, UK)
Joicy Xavier, PhD Student (UFMG, Brazil)
Edward Airey
Vittoria Cicaloni
Raid Halawani
Juliana Assis
Francislon Silv
Biography
Email:david.ascher [at] unimelb.edu.au ( )
|
Dr David Ascher is currently an NHMRC CJ Martin Fellow and Group Leader in the Department of Biochemistry and Molecular Biology, University of Melbourne. His research focus is in modelling biological data to gain insight into fundamental processes. One of his primary research interests has been developing tools to understand the molecular consequences of mutations on protein structure and function. This information has been used to unravel the link between genetic changes and phenotypes, enabling us to identify the changes that lead to genetic diseases and drug resistance. David has a B.Biotech from the University of Adelaide, majoring in Biochemistry, Biotechnology and Pharmacology and Toxicology; and a B.Sci(Hon) from the University of Queensland, majoring in Biochemistry, where he worked with Luke Guddat and Ron Duggleby on the structural and functional characterization of enzymes in the branched-chain amino acid biosynthetic pathway. David went to St Vincent’s Institute of Medical Research to undertake a PhD at the University of Melbourne in Biochemistry. There he worked under the supervision of Michael Parker using computational, biochemical and structural tools to develop small molecules drugs to improve memory. In 2013 David went to the University of Cambridge to work with Sir Tom Blundell on using fragment based drug development techniques to target protein-protein interactions; and subsequently on the structural characterisation of proteins involved in non-homologous DNA repair. He returned to Cambridge in 2014 to establish a research platform to characterise the molecular effects of mutations on protein structure and function- using this information to gain insight into the link between genetic changes and phenotypes. In late 2016, David was recruited to a lab head position in the Department of Biochemistry and Molecular Biology at the University of Melbourne, and established a lab at the Bio21 Institute. The research group is currently funded by several external grants. A list of David’s publications can be found via Google Scholar. David is an Associate Editor of Progress in Biophysics and Molecular Biology and a member of the External Academic Expert Board of the Tuscany University Network (TUNE) PhD Programme in Biochemistry. He holds positions at Sidney Sussex College, Cambridge, the Department of Biochemistry, University of Cambridge, and Centro de Pesquisas René Rachou, FIOCRUZ – Minas, Brazil |

